During the curing process of high alumina cement refractory castables or prefabs, there are often different degrees of pulverization and spalling.
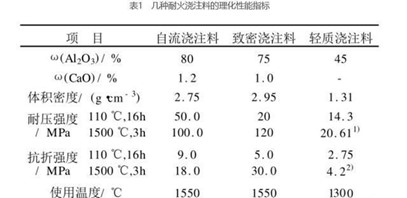
Table 1 lists several physical performance indicators of castables found in the actual field. The X-ray diffraction analysis of the white precipitates on the surface of the high alumina cement castable during the curing process shows that the main phase is Na2SO4, in addition, there are a small amount of corundum and mullite phases, corundum and molybdenum phases. The stone phase is the main phase of this high-alumina castable, which is brought in during sampling. The morphology of this powder was observed under an electron microscope, and it was found that most of the powders were irregular in shape, and a small amount had a ribbon-like helical structure.
The results of electron probe energy spectrum analysis show that the main components of the random-shaped powder are Na, S and O elements, as well as elements such as Al, Si, P, K, Ca and Fe. Compared with the X-ray diffraction pattern, it is determined to be Na2SO4. In the helical structure, in addition to the above elements, there are also a large amount of C and Cl elements, and the content of S element is low, and there may be chlorides and carbonates. Similarly, after immersing the pulverized and exfoliated powder of the superficial layer of the lightweight high alumina cement castable in water, the phase composition of the powder after filtration and crystallization is detected. The main phases are aqueous Na2SO4, anhydrous Na2SO4 and a small amount of Na2CO3.
The white precipitates on the surface of high alumina cement castables are of complex types, including soluble carbonates, sulfates, chlorides, etc. Ellson and Sakamoto et al. also found that the soluble precipitates are Na2CO3·nH2O (n=1,7,10 ). Although the reaction products of carbonation and sulfation may also include CaSO4·2H2O(s), Al(OH)3(s), CaCO3(s), Na2O·Al2O3, Na2O·11Al2O3, it can be considered that the actual surface of the castable The precipitates are mainly Na2CO3, Na2SO4, chlorides and other soluble salts, which are mainly related to the carbonation and sulfation process of the castable.
The acidic gases CO2, SO2, H2S in the air diffuse from the surface of the castable to the interior, and react with high alumina cement hydrate, and the products are Ca2CO3, CaSO4 2H2O(s), Al(OH)3(s) and other products. It is easy to precipitate in situ and does not aggregate with the migration of water molecules to the surface. The ions in the castable water such as Na+, K+, SO42-, CO32- and Cl-, etc., as the water molecules evaporate on the surface, the soluble compounds formed by their combination exceed the solubility in water and precipitate out. Further weathering and water loss, along with volume changes, will exacerbate the damage process.
For example, Na2CO3·nH2O and Na2SO4·nH2O are such soluble salts containing crystal water. The thermal analysis spectrum of the precipitates on the surface of the high alumina cement castable after dissolution and filtration shows that the main phase is Na2SO4·10H2O. It can be speculated that Na2SO4·10H2O is continuously crystallized from the aqueous solution, loses a part of crystal water at about 60℃, and further loses water and weathers to form Na2SO4 at about 100℃. In the process of dehydration and weathering of Na2SO4·10H2O, the volume shrinkage rate can reach 45.4% with the transformation of crystal form and volume shrinkage.
The acid gases CO2, SO2, and H2S in the air are one of the reasons for the carbonation and sulfation of high alumina cement castables, and the quality of water used for castables on site cannot be ignored, especially SO42- and H+ in water, which are very harmful to high alumina cement castables. Cement hydrates also play a certain erosive role.
